- Blog
- Home
- Splix Io Poki
- Jewel Quest Iii
- Battlezone Game
- Card Wars Adventure Time Online Game
- Warp 9
- Syberia 2 Download
- The Baconing Torrent
- Food Street Art
- Graceful Explosion Machine Switch
- Fishdom Depths Of Time Hints
- Silkroad Register
- Clustertruck Game Download
- Landmark Cinemas
- Madden NFL 16
- Shadow President Cheats
- Starlink Battle For Atlas Trailer
- The House Of Da Vinci Download
- Bridget Fonda
- Destruction Derby 2 Ps1
- Skip Boat
- City Builder Road Construction Videos
- Cake Mania Main Street For Mac
- Tiger Woods Pga Tour 12 The Masters Cheats
- Zombie Guard Dog
- Galactic Junk League Redeem Codes
- Armored Warriors Wiki
- My Talking Hank Game Play
- Sinistar Atari 2600
- Trackmania Turbo System Requirements
- Worms Open Warfare Ds
- Ascent The Space Game Uranium
- Red Baron Ii 3d
 
For molecules of the formula AX n, place the atom with the lower group number in the center. Place the atoms relative to each other.(For example, NO 3 - has 5 + 3圆 + 1 = 24 valence electrons NH 4 + has 5 + 4+1 – 1 = 8 valence electrons.) (For example, H 2O has 2x1 + 6 = 8 valence electrons, CCl 4 has 4 + 4x7 = 32 valence electrons.) For anions, add one valence electron for each unit of negative charge for cations, subtract one electron for each unit of positive charge. Count the total number of valence electrons in the molecule or polyatomic ion.It’s very simple, just put the name of the compound or chemical formula of the compound in the blank box of the “Lewis structure generator” tool, then, click on submit, and a popup box will appear with the Lewis structure of the given compound. How do you use the Lewis structure calculator tool? Lewis structure or Lewis dot structure is drawn by using 5 or 6 steps. Let’s check our full guide in – How to draw the lewis structure for any compound or ions?Īlso, check the Lewis structure for some important molecules – CO2 Lewis structure That’s all by working on the methods given above, you can calculate the lewis structure for any compound. 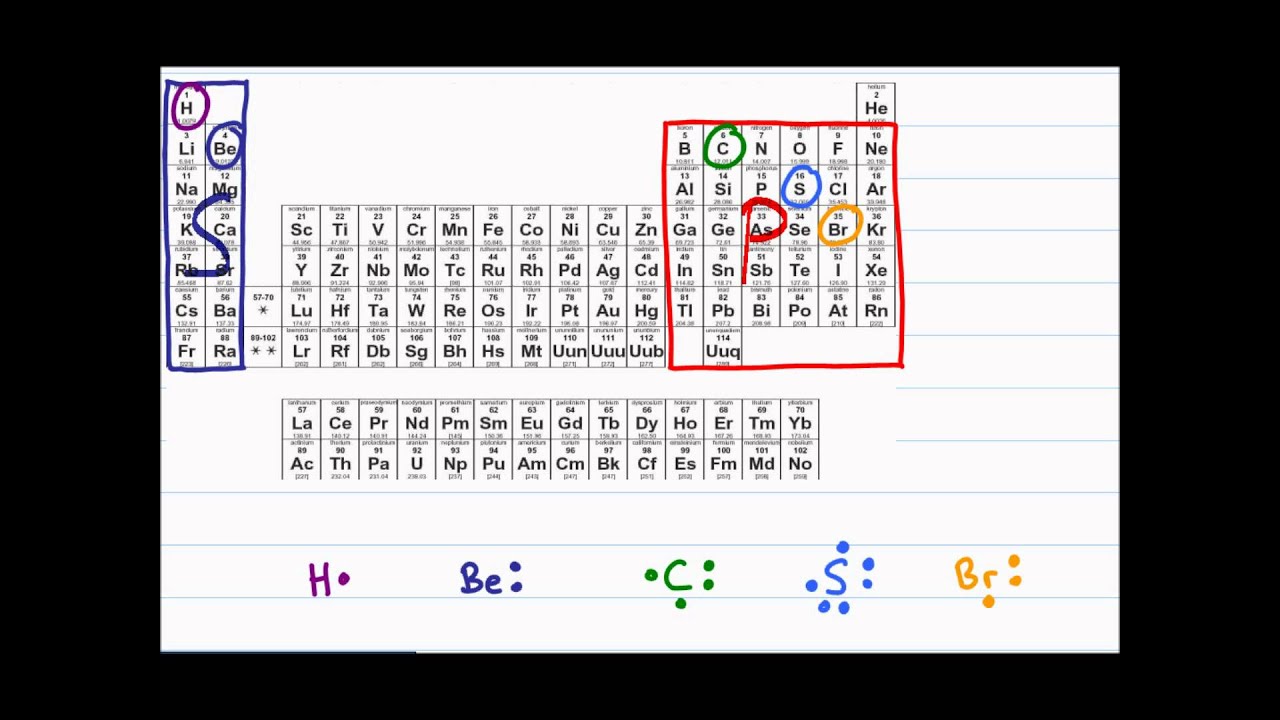
So, an easy way to find the valence electron of atoms in the PCl 3 molecule is, just to look at the periodic group of phosphorous and chlorine atoms. Steps for drawing the Lewis dot structure for PCl 3įirst of all, determine the valence electron that is available for drawing the lewis structure of PCl 3 because the lewis diagram is all about the representation of valence electrons around atoms. 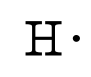
Let’s follow some steps and draw the lewis structure for PCl3. For that, either you can use the online “Lewis structure calculator” tool or the traditional method. Complete the octet of the central atom (make multiple bonds if necessary)įor example – Let’s say we have to calculate the lewis structure for the PCl3 molecule.Complete the octet of outer atoms first.Calculate the total number of valence electrons in the molecule.For most of the compounds, the lewis structure is drawn using 5 or 6 simple steps. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
